訪問マッサージはどのような人におすすめ?訪問マッサージの関連情報を紹介
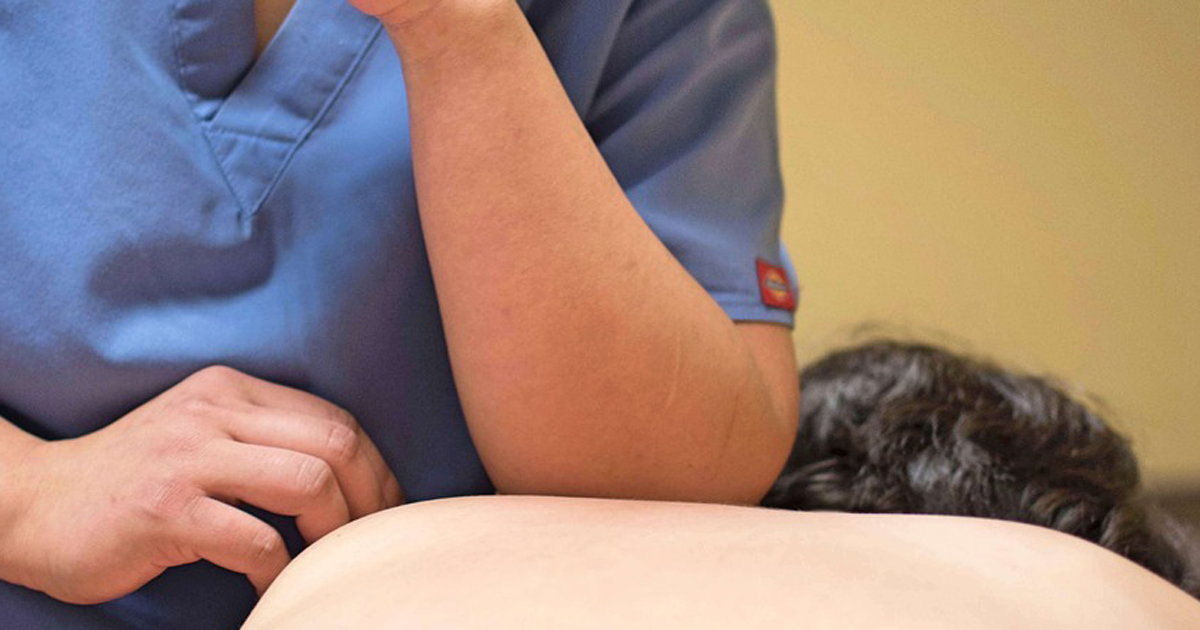
訪問マッサージでは、症状に応じた施術を受けることが可能です。
例えば肩こりがひどく悩まされいるなら、首や肩甲骨周りをほぐすマッサージを受けることができます。
訪問マッサージでは肩だけでなく全身にアプローチして、症状の軽減を目指します。
利用する前に関連情報をある程度把握しているなら、心の準備ができるにちがいありません。
訪問マッサージでは病気がすぐに治ると言うわけではありませんが、体をほぐし血行を良くするので気分が良くなります。
-
訪問マッサージはどのような人におすすめ?訪問マッサージの関連情報を紹介
- 寝たきりの方は訪問マッサージを利用するべき
- 手足の筋肉に麻痺がある場合は訪問マッサージがおすすめ
- 訪問マッサージは施術だけではなくおしゃべりも楽しめる
- 訪問マッサージは筋肉の深部にある神経へ直接アプローチできる
- 訪問マッサージは患者さんと信頼関係を築くのが大事(2024.4.1)
- 訪問マッサージは1回あたりの自己負担額が少ない(2024.4.1)
- 訪問マッサージは長時間待たなくてもOK(2024.4.1)
- 心身ともに健康な毎日を目指すなら訪問マッサージ(2024.4.1)
- 訪問マッサージは歩行困難な方におすすめ(2024.4.1)
寝たきりの方は訪問マッサージを利用するべき
寝たきりの方が訪問マッサージを利用すべき理由は多岐にわたります。
まず第一に身体を動かすことが難しくなります。
その結果、血液やリンパの流れが滞り、筋肉の衰えや肩こり、腰痛などの身体の不調が起こりやすくなります。
専門のマッサージ師が自宅などに訪れてマッサージを行うため、寝たきりの方でも身体を動かすことなくマッサージの恩恵を受けることができます。
第二に長時間同じ姿勢で過ごすため、体の一部が圧迫されることで褥瘡(じょくそう)と呼ばれる皮膚の潰瘍ができるリスクが高まります。
マッサージ師が適切な体位を提案し、圧迫を軽減する施術を行うことで、褥瘡予防にもつながります。
日常的なストレッチや運動が難しいため、筋肉の萎縮や関節の硬直が進む可能性があります。
マッサージ師が筋肉や関節をほぐすことで、筋力や関節の可動域を改善し、機能回復を促進します。
社会との交流が限られがちで孤立感を抱えることがあります。
訪問マッサージは、定期的な訪問によって患者とのコミュニケーションを促進し、心のケアにも寄与します。
訪問マッサージは家族や介護者にとっても負担を軽減する点があります。
訪問マッサージ師が専門的な施術を行うことで、家族や介護者は身体のケアに専念することができ、心身ともに休息を取る時間を確保できます。
手足の筋肉に麻痺がある場合は訪問マッサージがおすすめ
麻痺が手足に現れると日常生活において多くの制約が生じます。
そのような状況で訪問マッサージは有益な選択肢と言えます。
訪問マッサージはマッサージ師が自宅や施設を訪れて個別に施術を行うサービスです。
利点は便利さといえるでしょう。
外出することが困難であることがあります。
そのため自宅や施設で施術を受けることができる訪問マッサージは、患者や家族にとって大変助かるものとなります。
外部の施設に行く必要がないためストレスを軽減し、リラックスした状態でマッサージを受けることができます。
個別の施術が受けられる点も挙げられます。
1対1で行われるため、マッサージ師は患者の状態やニーズに応じた施術を提供できます。
筋肉の弛緩や血行促進が必要とされることがありますが、一般的なマッサージとは異なるアプローチが求められます。
そのようなニーズに応えることができるため、より効果的な治療が期待できます。
患者の自立支援にも繋がります。
麻痺によって日常生活動作に制限が生じる場合、患者は依存度が高まりがちですが、定期的なマッサージによって筋肉の機能が改善されることで、自己介助が可能になる場合があります。
自立した生活をサポートすることで、心身の健康を維持し生活の質を向上させることができます。
訪問マッサージは施術だけではなくおしゃべりも楽しめる
訪問マッサージは身体の悩みを解消するだけでなく、おしゃべりを楽しむこともできます。
あん摩マッサージ指圧師の施術を受けながら、いろいろな会話を楽しめるのです。
会話に意味を持たせる必要はなく、とりとめのない話だけでも問題ありません。
言葉を交わすこと自体が効果的なコミュニケーションになるからです。
自分の気持ちを言葉として吐き出すことで、心が軽くなっていくのがわかるでしょう。
寝たきりになってしまった人、身体が不自由で外出が難しい人などは、ストレスを溜め込みやすい傾向があります。
対策をせずにストレスを放置すると、次第に心が蝕まれていきます。
特にひとり暮らしをしている人は、ストレスのはけ口を見つけにくいです。
そこで訪問マッサージを活用して、定期的におしゃべりを楽しみましょう。
和気あいあいとした雰囲気の中で会話を楽しむためには、あん摩マッサージ指圧師との相性がポイントになります。
自分と価値観がマッチする、なんとなく居心地がよい、などを目安に探してみてください。
訪問マッサージは筋肉の深部にある神経へ直接アプローチできる
訪問マッサージは、筋肉の深部にある神経へ直接アプローチできる効果的なリラクゼーション法です。
この施術は専門的な技術を持つマッサージセラピストによって行われます。
通常、日常の活動やストレスによって緊張し硬くなりがちです。
これにより深部にある神経にも圧迫や緊張が生じることがあります。
訪問マッサージは、そうした筋肉の問題に直接届けることができます。
マッサージセラピストは特定のグループや問題箇所を的確に把握し、適切な手技を用いて施術を行います。
圧をかけることで緩め血流を促進することで栄養や酸素を筋肉に供給し、老廃物を排出する働きがあります。
これにより緊張が解消され、神経にかかる負担が軽減されます。
訪問マッサージは、自宅やオフィスなどで受けることができる利便性が魅力的です。
また、個別のニーズに合わせたカスタマイズされた施術が可能であり、リラクゼーションだけでなく、疲労回復やストレス軽減にも効果が期待されます。
訪問マッサージは患者さんと信頼関係を築くのが大事
マッサージを受けることで、体の凝りや疲れを癒してもらうことができますが、わざわざ店舗を構えているところに出向くことが出来ない場合もあります。疲れすぎて立ち寄ることができなかったり、マッサージの施術を受けた後はすっきりするので体の力が抜けて、そこから自宅まで帰宅することが面倒だということもあるでしょう。そんな時は訪問マッサージを利用すれば、自宅や宿泊している施設などに出向いてもらうことができるので、出向く手間を省けます。出張や旅行などで慣れない場所に行くと、いつもより疲れることがあるので、そんな時にも訪問マッサージは最適です。ただ訪問マッサージ重要なことは、施術するスタッフさんと患者さんとの信頼関係が重要だということになります。人間には相性というものが大事なので、いくら施術のスキルが高くても性格的に合わないということもありますから、まずは信頼関係を築くことが欠かせません。もし信頼を構築できれば、リピーターになってくれるので、仕事の売り上げもアップします。
訪問マッサージは1回あたりの自己負担額が少ない
健康保険が適用される訪問マッサージには、1回あたりの自己負担額が比較的少ない、という大きな利点があります。外部の施設にマッサージのために通院するという場合には、診療費用以外にも交通費や駐車料金など、様々なお金がかかるものです。交通の便があまりよくない地域に住んでいる場合、遠方まで外出するためには、色々と手間がかかります。公共交通機関を利用する場合はかなりの交通費が必要になりますし、自家用車などで移動する場合も、ガソリン代や駐車費用がかさんでしまうものです。自宅まで来てもらうことができる訪問マッサージなら、そうした諸費用も節約することができます。訪問マッサージの料金体系は、施術内容や時間帯によって柔軟に設定されていますので、予算に合わせて利用することが可能です。訪問マッサージは、経済的に利用することができる非常に便利なサービスだといえます。経済的な負担を最小限に抑えつつ不調を改善したい、という場合に最適だといえるでしょう。
訪問マッサージは長時間待たなくてもOK
店舗型のマッサージ店では複数のお客さんが訪れることから、順番待ちが発生することがあります。予約をしていたとしても急な体調不良やアクシデントによる怪我などから飛び込みで来た緊急性の高い方を優先することもあり、順番が繰り下げられてしまうことも少なくありません。その一方で訪問マッサージの場合はあらかじめ予約をしておいた日時に指定した場所へスタッフが訪問することから、長時間待たなくても良いのが評判です。訪問マッサージは実店舗を持たず事務所とスタッフの待機所のみで運営されていることが多く、飛び込みのお客さんが間に入ることが無いのはもちろんのこと、予約が確定された段階で担当するスタッフが来てくれるのが確実になります。急に待ち時間が発生したり後回しにさせることが一切ないことから、自身のタイミングで施術を受けることができます。このような体制が整っているのも訪問マッサージの魅力で、根強いファンが居るのも頷けるところです。
心身ともに健康な毎日を目指すなら訪問マッサージ
健康をずっと維持するなら、心身の不調を防ぐことが大切です。訪問マッサージは身体の機能を復活させるのが目的ですが、健康な人でも利用できます。自由診療の訪問マッサージは保険診療の制限を受けないので、自分の好きなときに好きなメニューを受けられます。あん摩マッサージ指圧師の施術は、身体のバランスを調整するのに最適です。病気は心からと言われるように、特に心の状態が不安定になっているときは要注意です。訪問マッサージは身体をほぐしつつ、心のわだかまりも取り除いてくれます。施術後は身体が軽くなるだけでなく、脳が元気になるという人が少なくありません。訪問マッサージには病気予防の側面もあるので、心身を健康にするためにも活用してください。一般的な利用頻度は週に1~数回になりますが、健康な方であれば月に1回でも問題ありません。コリが気になるとき、ストレスが抜けないとき、脳がすっきりしないときなどに利用すれば病気の予防にもなります。
訪問マッサージは歩行困難な方におすすめ
訪問マッサージは歩行困難な方にとって特に有益なサービスとなります。店舗への足を運ぶことが難しい方でも自宅や指定した場所で専門的なマッサージを受けることができます。 歩行困難な方は日常生活での移動が困難であり、それにより筋肉が硬くなりやすい傾向にあります。定期的なマッサージは筋肉の硬化を和らげ血行を改善し、さらに疲労感を軽減します。また、筋肉の柔軟性を保つことは日常生活での動作を支え、身体のバランスを保つ上で重要です。 また、訪問サービス心地よいリラクゼーションを提供するとともに孤独感の軽減にも役立つ可能性があります。人との直接的な接触は心身の健康を促進する効果があると広く認知されています。訪問マッサージの専門家とのコミュニケーションは、心地よい人間関係を築き、精神的な安定を支える一助となります。 さらに、訪問マッサージのプロフェッショナルは適切な施術とアドバイスにより健康状態の改善や維持をサポートします。これにより長期的な生活の質の向上に寄与することが期待できます。 つまり、訪問マッサージは、歩くのが難しい方が安心して身体と心のケアを受けることができる、非常に有益なサービスと言えるでしょう。